We move from the electromagnetic fields of induction to the biopolymer gelation of Spherification. Popularized by the modernist kitchen of Ferran Adrià, this technique allows a chef to encapsulate a liquid inside a thin, transparent gel membrane that “pops” upon consumption. It is a study in ion-exchange cross-linking, where the physical state of a liquid is manipulated by triggering a rapid phase change at the molecular surface.
To master spherification, one must understand the relationship between polysaccharide chains and divalent cation bridging.
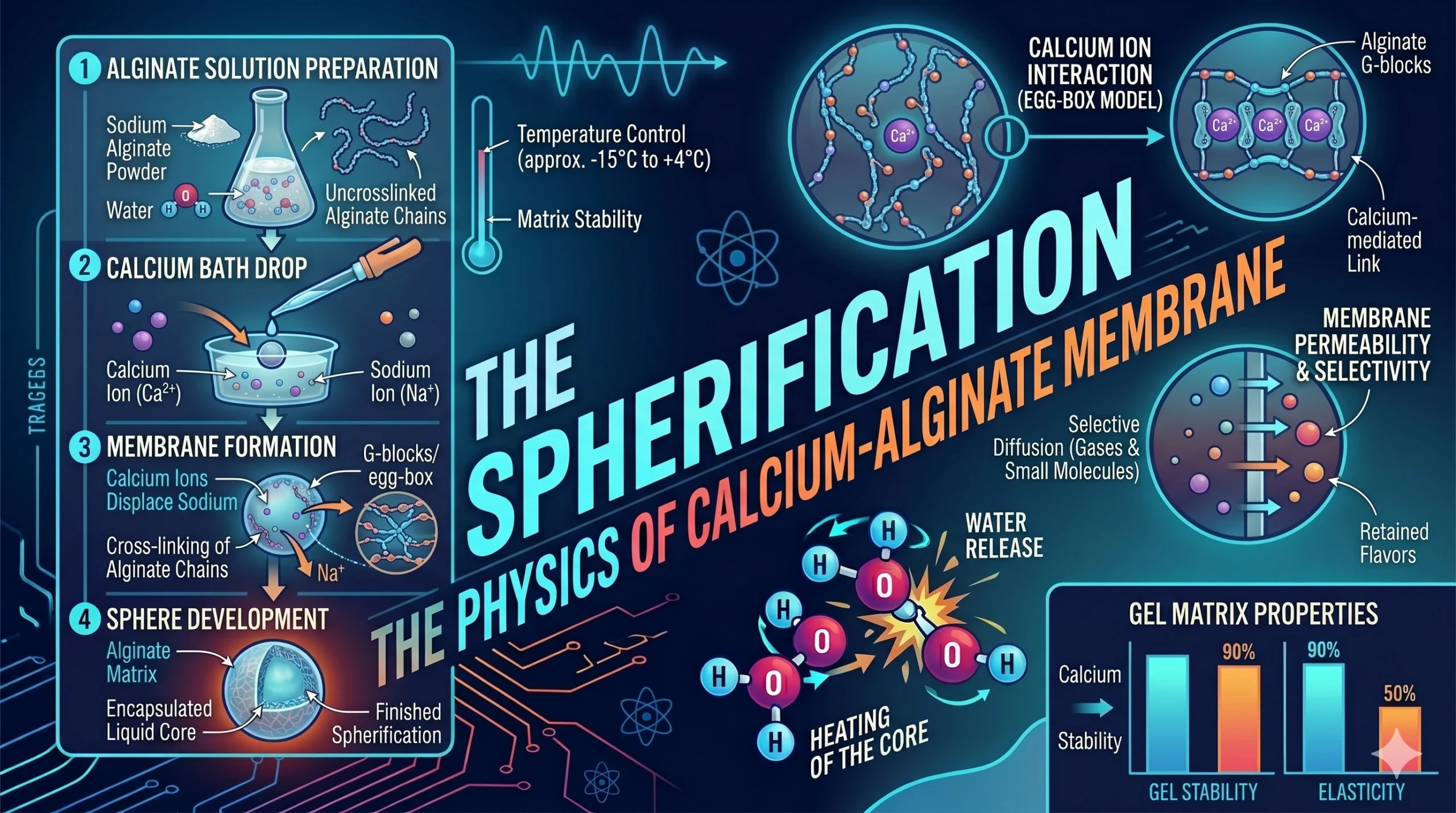
Part 1: Sodium Alginate – The Linear Polysaccharide
The core material used in spherification is Sodium Alginate, a natural polymer extracted from brown seaweed.
- Molecular Structure: Alginate consists of long, linear chains of carbohydrate molecules. In its sodium form, these chains repel each other, allowing the alginate to dissolve into a liquid and flow freely.
- The “Liquid State”: At this stage, the liquid is viscous but has no structural integrity. It behaves like a thick syrup.
Part 2: Ion Exchange – The Calcium Trigger
The transformation from liquid to gel occurs when the alginate comes into contact with Calcium ions ($Ca^{2+}$).
- Divalent Bridging: Unlike sodium ($Na^{+}$), which has a single positive charge, calcium is a divalent cation with two positive charges. This allows a single calcium ion to act as a “bridge” between two different alginate chains.
- The “Egg Box” Model: As calcium ions displace the sodium ions, they pull the alginate chains together into a tight, three-dimensional grid. This structure is known as the “egg box” model, resulting in a solid, elastic hydrogel membrane.
Part 3: Basic vs. Reverse Spherification – Managing the Diffusion
Depending on where the calcium and alginate are placed, the chef can control the membrane growth.
- Basic Spherification: The alginate is added to the flavored liquid and dropped into a calcium bath. The membrane grows inward. This is ideal for small “caviar” pearls, but the gelation continues until the entire sphere becomes a solid jelly ball.
- Reverse Spherification: The calcium is added to the flavored liquid (or is naturally present, like in dairy) and dropped into an alginate bath. The membrane grows outward. Because the alginate cannot penetrate the already-formed gel, the center remains liquid indefinitely. This is the gold standard for large “spheres” or “ravioli.”
Conclusion: The Engineering of Encapsulation
Spherification proves that texture can be a boundary layer. By leveraging the specific reactive properties of seaweed polymers and calcium ions, the chef creates a vessel out of the ingredient itself. It is the physics of surface-level gelation—a way to package flavor in a fragile, temporary architecture.