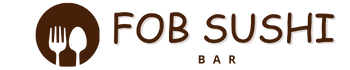Introductory sub headline
This is placeholder text which will be replaced by your content. Lorem ipsum amet elit morbi dolor tortor. Vivamus eget mollis nostra ullam corper. Pharetra torquent auctor metus felis nibh velit tellus semper taciti nostra. Semper pharetra montes habitant congue integer magnis nulla.
Aerated Foams – The Physics of Surfactant Surface Tension
We move from the encapsulated liquids of spherification to the fluid mechanics of Aerated Foams. In the modern kitchen, a foam...
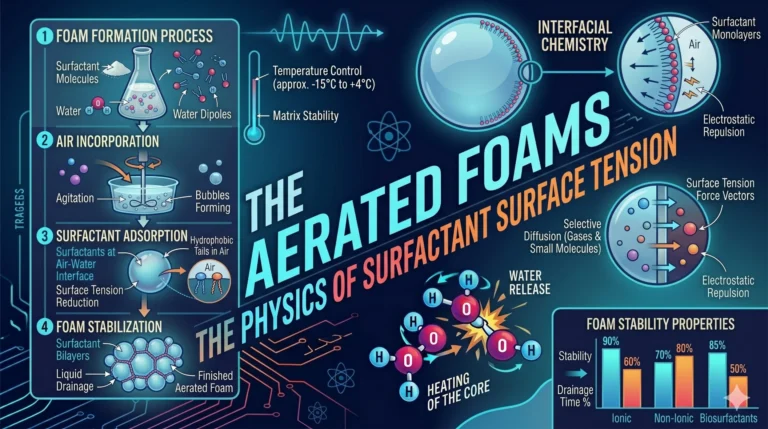
Read MoreSpherification – The Physics of the Calcium-Alginate Membrane
We move from the electromagnetic fields of induction to the biopolymer gelation of Spherification. Popularized by the modernist kitchen of Ferran...
Read MoreThe Induction Cooktop – The Physics of Magnetic Flux and Eddy Currents
We move from the molecular structure of gelatin to the electromagnetic engineering of the Induction Cooktop. Unlike gas or traditional electric...
Read MoreGelatin Clarification – The Physics of the “Freeze-Thaw” Filter
We move from electromagnetic rotation to the structural thermodynamics of Gelatin Clarification. While the Consommé (Article 65) uses a “raft” for...
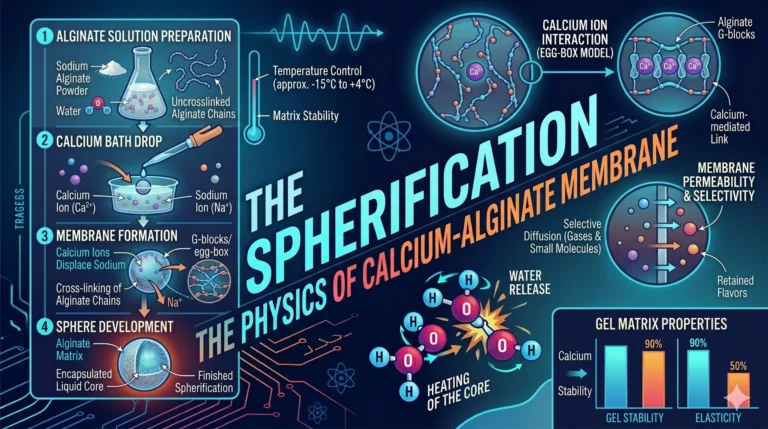
Read MoreThe Microwave – The Physics of Dipolar Rotation
We move from the high-pressure conduction of the pressure cooker to the electromagnetic engineering of the Microwave. Often misunderstood as “cooking...
Read MoreThe Pressure Cooker – Atmospheric Pressure and Boiling Point Elevation
We move from the infusion of gas into solids to the high-pressure thermodynamics of the Pressure Cooker. This tool is the...
Read MoreSiphon-Carbonated Fruit – The Physics of $CO_2$ Infusion
We move from the molecular browning of the Maillard reaction to the high-pressure thermodynamics of Siphon Carbonation. This technique allows a...
Read MoreThe Maillard Reaction – The Molecular Chemistry of Browning
We transition from the structural engineering of meat to the fundamental chemical engine behind almost every savory flavor in the kitchen:...
Read MoreThe Emulsified Sausage – The Engineering of the “Primary Bind
We shift from the crystalline structure of chocolate to the biopolymer engineering of the Emulsified Sausage (such as Mortadella, Frankfurters, or...
Read MoreChocolate Tempering – The Physics of the Type V Crystal Lattice
We move from the filtration of savory liquids to the solid-state physics of Chocolate Tempering. Chocolate is a polymorphic substance, meaning...
Read MoreSign up for our email list
This is placeholder text which will be replaced by your content.
Email Address
*Replace this mock form with your preferred form plugin