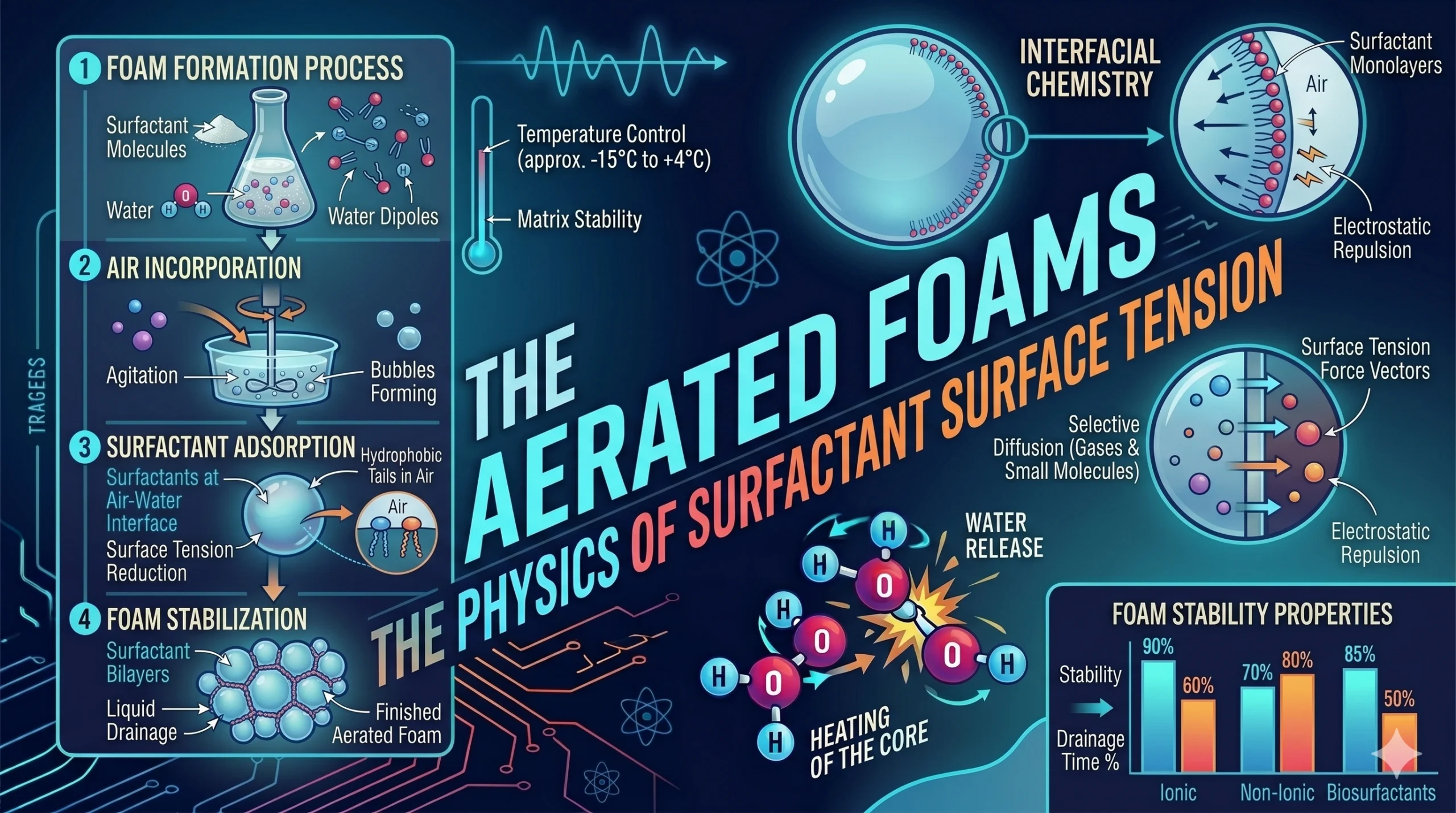
We move from the encapsulated liquids of spherification to the fluid mechanics of Aerated Foams. In the modern kitchen, a foam is not just a garnish; it is a colloidal dispersion where a gas is trapped within a liquid or solid matrix. By manipulating surface tension and utilizing surfactants, a chef can engineer textures ranging from “airs” that vanish on the tongue to dense, stable mousses that hold their shape for hours.
To master aerated foams, one must understand the relationship between amphiphilic molecules and Laplace pressure.
Part 1: Surfactants – The Molecular Bridge
At the interface of air and water, surface tension acts as a barrier, causing bubbles to collapse as they seek the lowest energy state. To stabilize a foam, a chef must introduce a surfactant (such as lecithin, egg proteins, or monoglycerides).
- Amphiphilic Nature: Surfactants are “bilingual” molecules; they have a hydrophilic (water-loving) head and a lipophilic (fat-loving/air-seeking) tail.
- The Protective Film: When air is whisked into the liquid, the surfactants align themselves at the boundary. The heads stay in the water while the tails point into the air bubble. This reduces the surface tension, preventing the bubbles from merging (coalescence).
Part 2: The Siphon Whip – Henry’s Law and Rapid Expansion
While whisking creates a foam through mechanical agitation, the iSi Siphon (Whipping Siphon) uses dissolved gas to engineer aeration from the inside out.
- Pressure Loading: A gas—usually Nitrous Oxide ($N_2O$)—is charged into a sealed container. Because $N_2O$ is highly lipophilic, it dissolves into the fats and liquids of the base.
- Instantaneous Nucleation: When the trigger is pulled, the pressure drops to atmospheric levels. The dissolved gas instantly expands, creating millions of microscopic bubbles simultaneously throughout the entire volume of the liquid. This results in a foam with much smaller, more uniform bubbles than mechanical whisking can achieve.
Part 3: Foam Stability – Drainage and Ostwald Ripening
The “life” of a foam is a battle against two physical forces: Drainage and Ostwald Ripening.
- Drainage: Gravity pulls the liquid down through the bubble walls (the lamellae). If the liquid drains too fast, the walls become thin and pop. Stabilizers like Xanthan Gum are often added to increase the viscosity of the liquid, slowing the drainage rate.
- Ostwald Ripening: This is the process where small bubbles shrink and large bubbles grow due to differences in internal pressure (Laplace Pressure). A successful culinary foam uses proteins or fats to create a rigid enough skin to resist this pressure-driven migration of gas.
Conclusion: The Architecture of Nothing
Aerated foams prove that volume can be decoupled from mass. By leveraging the surfactant properties of lipids and proteins, the chef engineers a structure that is $90\%$ air but $100\%$ flavor. It is the physics of interfacial stabilization—a way to transform a heavy sauce into a weightless cloud.