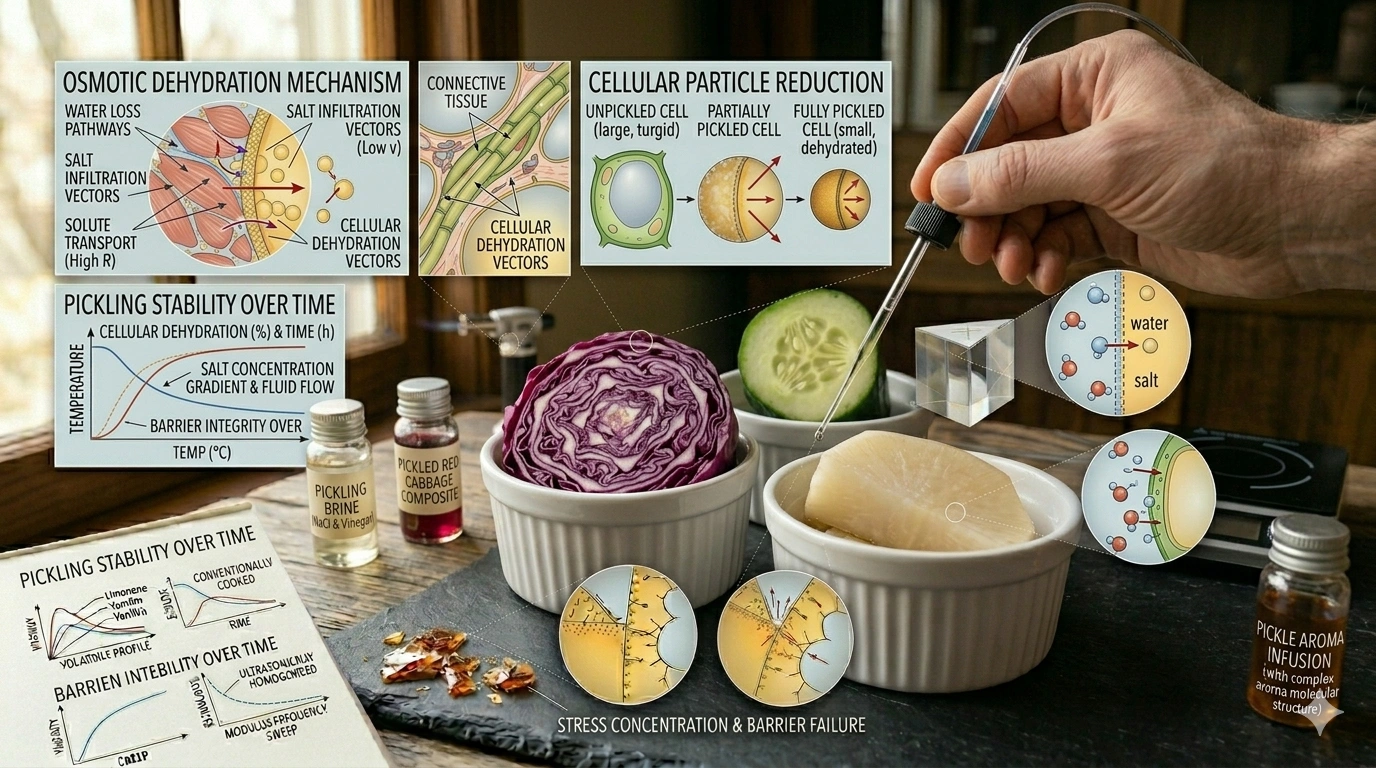
We move from the thermal expansion of foams to the biochemical engineering of the Osmotic Pickle. Pickling is often viewed as a simple preservation method, but it is actually a study in fluid dynamics and selective permeability. By manipulating the concentration of solutes in a brine, a chef can trigger a massive migration of water across cell membranes, fundamentally altering the texture and “crunch” of a vegetable through the management of turgor pressure.
To master the Osmotic Pickle, one must understand the relationship between osmotic potential and the hemicellulose structure of plant cell walls.
Part 1: Osmosis – The Drive for Equilibrium
At the heart of every pickle is Osmosis: the movement of solvent (water) from an area of low solute concentration to an area of high solute concentration through a semi-permeable membrane.
- The Gradient Force: When a cucumber is placed in a high-salt brine, the concentration of salt outside the vegetable is significantly higher than inside. Nature seeks equilibrium, forcing the water inside the vegetable cells to rush outward to dilute the surrounding brine.
- Cellular Dehydration: As water exits the cells, the internal pressure—known as turgor pressure—drops. In the short term, this makes the vegetable limp, but it is the essential first step in creating a space for the flavored brine to eventually re-enter the tissue
Part 2: The Stiffening Phase – Pectin and Calcium Ions
A common technical failure in pickling is “mushiness.” This is caused by the breakdown of pectin, the glue that holds plant cells together.
- Pectin Methylesterase (PME): By maintaining the initial brine temperature between $50^{\circ}C$ and $70^{\circ}C$, the chef activates PME. This enzyme alters the pectin molecules so they can more easily bind with minerals.
- Calcium Cross-Linking: Often, chefs add a source of calcium (like calcium chloride) to the brine. The calcium ions act as molecular “bridges” between pectin chains, reinforcing the cell walls and ensuring the pickle retains a “snap” even after long-term storage.
Part 3: Lactic Acid Fermentation – The Biological Acidification
While quick-pickles use vinegar (acetic acid), traditional fermented pickles rely on Lactic Acid Bacteria (LAB) to engineer the environment.
- The Selective Barrier: Salt serves a dual purpose. It draws out water and sugars via osmosis, but it also creates a toxic environment for “bad” spoilage bacteria. LAB, however, is salt-tolerant.
- The pH Shift: As the LAB consumes the sugars drawn out by osmosis, it produces lactic acid as a byproduct. This lowers the pH of the brine to below $4.6$. This acidity not only preserves the food but also denatures certain proteins, creating a unique “bright” flavor profile that cannot be replicated with added vinegar.
Conclusion: The Engineering of Turgor
The Osmotic Pickle proves that preservation is a mechanical process. By using salt to drive fluid migration and enzymes to reinforce cellular architecture, the chef transforms a perishable vegetable into a structurally sound, shelf-stable ingredient. It is the physics of solute-driven transformation.